How to Read a Certificate of Analysis: Evaluating Peptide Quality Documentation
A Certificate of Analysis is either evidence of quality or a meaningless document. Understanding the difference protects research integrity.
In an unregulated market, the COA serves as the primary quality verification mechanism. However, not all COAs represent equivalent rigor. Some document genuine analytical testing from independent laboratories. Others are fabricated documents with invented data.
This guide explains how to evaluate COA legitimacy and interpret analytical results.
Essential COA Elements
A legitimate Certificate of Analysis contains several required elements:
1. Product Identification
- Product name: The specific peptide (e.g., “BPC-157 Acetate”)
- Batch/Lot number: Unique identifier enabling traceability
- Molecular formula: Chemical composition (e.g., C₆₂H₉₈N₁₆O₂₂)
- Molecular weight: Expected mass (e.g., 1419.53 Da)
- CAS number: Chemical Abstracts Service registry number, if applicable
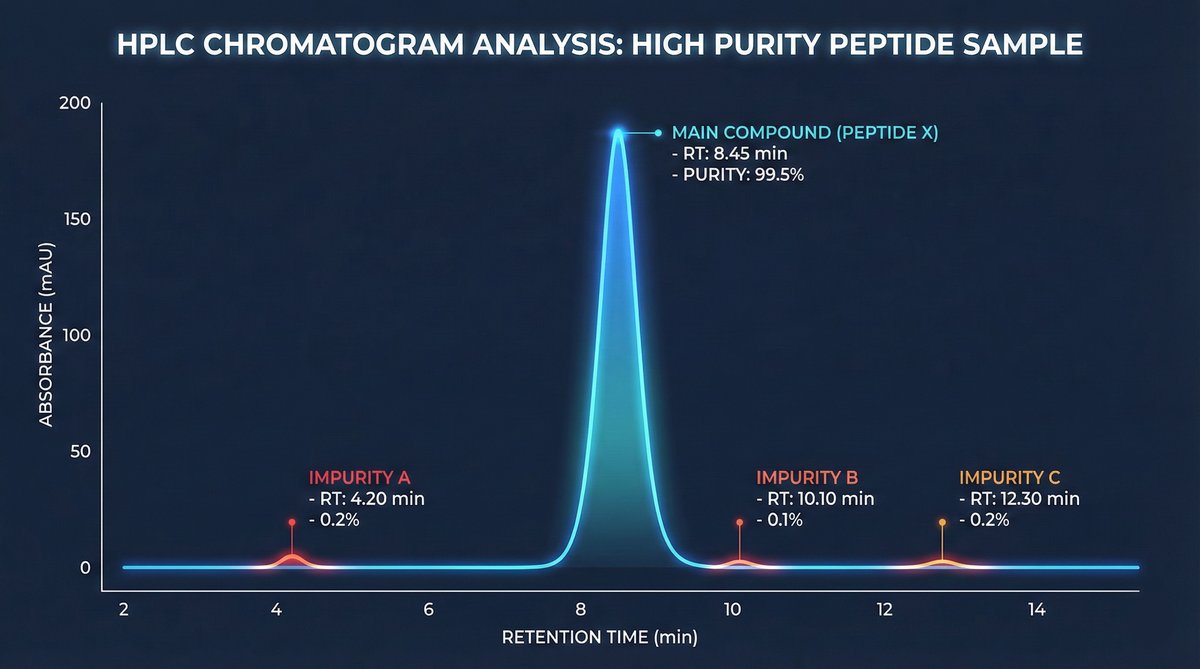
2. Purity Analysis (HPLC)
High-Performance Liquid Chromatography results should include:
- Purity percentage: Stated clearly (e.g., “99.2%”)
- Method description: Column type, mobile phase, detection method
- Chromatogram: The actual graph showing peaks and their relative areas
- Retention time: When the main peak eluted from the column
Interpretation: Purity exceeding 99% indicates research-grade material. Below 95% is questionable for research applications. Below 90% represents unacceptable quality regardless of price.
3. Identity Confirmation (Mass Spectrometry)
Mass Spec results should include:
- Observed molecular weight: The actual measured mass
- Expected molecular weight: The target value
- Mass spectrum: Graph showing mass/charge peaks
- Deviation: Difference between observed and expected values
Interpretation: Observed mass should match expected mass within instrument tolerance (typically ±0.1%). Significant deviation suggests incorrect product or contamination.
4. Laboratory Information
- Laboratory name: The organization that performed testing
- Testing date: When analysis occurred
- Analyst signature or ID: Individual accountability
- Contact information: Verification capability
5. Physical Characteristics
- Appearance: Expected form (e.g., “white lyophilized powder”)
- Solubility: Behavior in relevant solvents
- Storage conditions: Recommended handling parameters
Warning Signs: Indicators of Questionable COAs
🚩 No Laboratory Identification
COAs without named testing laboratories likely represent in-house generated documents. When vendors grade their own products, the conflict of interest is structural. Independent verification requires independent verifiers.
🚩 No Chromatogram or Mass Spectrum
A document stating “Purity: 99%” without showing analytical data is an assertion, not evidence. The graphs prove analysis was actually performed. Anyone can type numbers; analytical data requires actual testing.
🚩 Missing Batch Number
No batch number means no traceability. Quality control requires the ability to identify which production run generated specific products. Missing batch numbers suggest inadequate quality management systems.
🚩 Generic Template Appearance
Legitimate analytical reports from professional laboratories have consistent, standardized formatting. COAs that appear to be Word documents with basic formatting warrant skepticism.
🚩 Purity Claims Without Method Details
Stating “99% pure” without methodology is meaningless. What HPLC column? What mobile phase? What detection wavelength? Legitimate laboratories document these parameters.
🚩 Identical COAs Across Different Batches
Each production batch should have unique testing documentation with its own batch number and testing date. Identical COAs for products purchased months apart indicate testing isn’t performed per-batch.
🚩 Unverifiable Laboratory References
Legitimate analytical laboratories have verifiable existence—websites, addresses, phone numbers. If the named laboratory cannot be found through standard searches, the COA may be fabricated.
COA Verification Process
Step 1: Verify laboratory existence. Search for the named laboratory. Professional analytical labs maintain web presence with contact information. Inability to find evidence of the laboratory’s existence indicates potential fabrication.
Step 2: Check for accreditations. Legitimate testing laboratories often hold ISO 17025 accreditation or similar credentials. Accreditation doesn’t guarantee honesty but indicates professional standards and external auditing.
Step 3: Examine the chromatogram. Does it appear to be genuine analytical data? The main peak should be sharp and well-defined. Multiple large peaks indicate significant impurities. Baseline noise patterns should appear natural, not artificially smooth.
Step 4: Verify molecular weight. The observed molecular weight should match published values for the specified peptide. PubChem provides expected masses for comparison.
Step 5: Contact the laboratory. For critical purchases, contact the testing laboratory directly to verify batch number and results. Legitimate laboratories will confirm their reports; fabricated COAs will not be recognized.
Understanding “Purity Exceeding 99%”
When Epiq Aminos specifies purity exceeding 99%, this means:
The main peak area on HPLC chromatography represents more than 99% of total detected material. The remaining <1% may consist of:
- Minor synthesis byproducts
- Trace degradation products
- Residual solvents at safe levels
This represents research-grade purity—appropriate for scientific investigation where observed effects must be attributable to the target compound, not contaminants.
Compare to vendors offering 80-90% purity: 10-20% of the material is not the labeled compound. In research contexts, that represents a substantial uncontrolled variable.
Epiq Aminos COA Standards
Every product includes a Certificate of Analysis documenting:
- ✓ Third-party laboratory testing (independent, not in-house)
- ✓ HPLC chromatogram showing purity exceeding 99%
- ✓ Mass spectrometry confirmation of molecular identity
- ✓ Unique batch number for production lot traceability
- ✓ Testing date and laboratory identification
COAs are accessible on product pages. If documentation isn’t visible for a specific product, contact us—we’ll provide it. Products without available COAs don’t ship.
The Standard for Research Quality
In an unregulated market, the COA represents the primary evidence of quality. Understanding how to evaluate these documents protects research integrity and distinguishes legitimate suppliers from those selling unverified materials.
Epiq Aminos COAs are available for inspection. Testing laboratories can be contacted for verification. Batch numbers enable traceability. This transparency is the foundation of trust in a market without regulatory oversight.
Questions about COA interpretation or specific product documentation? Contact us.
All products are intended for in-vitro research and educational purposes only. Not for human or veterinary use.