GLP Receptor Agonists: Molecular Mechanisms and Research Applications
Ozempic. Wegovy. Mounjaro. Zepbound.
These brand names represent a class of peptides that has generated significant scientific interest. GLP receptor agonists are among the most actively researched peptide therapeutics in modern pharmacology, with extensive published literature across multiple peer-reviewed journals.
Understanding the molecular biology of GLP receptor agonists explains why this compound class has become a major focus of metabolic research worldwide.
The GLP System: Molecular Mechanisms
GLP stands for Glucagon-Like Peptide. These are endogenous hormones produced by L-cells in the intestine in response to nutrient intake. The GLP signaling system regulates:
- Satiety signaling — GLP-1 receptors in the hypothalamus modulate appetite
- Glucose homeostasis — Stimulates insulin secretion, suppresses glucagon release
- Gastric motility — Delays gastric emptying, extending nutrient absorption time
- Reward pathway modulation — Affects mesolimbic dopamine circuits related to food reward
GLP receptor agonists are synthetic peptides engineered to activate these receptors with greater potency and duration than endogenous hormones. The molecular modifications that extend half-life from minutes to days represent significant peptide engineering achievements.
The Major Compounds: Molecular Profiles
Semaglutide (GLP-1 Agonist)
Brand names: Ozempic® (diabetes indication), Wegovy® (weight management indication)
Manufacturer: Novo Nordisk
Semaglutide is a 31-amino-acid peptide (molecular weight: 4,113.58 Da) with a C18 fatty acid side chain that enables albumin binding, extending plasma half-life to approximately 7 days. This allows once-weekly administration in clinical settings.
The clinical trial data is extensive. The STEP clinical trial program has been published in peer-reviewed journals including The New England Journal of Medicine (PMID: 33567185). The SELECT cardiovascular outcomes trial evaluated semaglutide in patients with established cardiovascular disease and has been published in NEJM. These trials enrolled tens of thousands of participants across multiple Phase 3 protocols.
Tirzepatide (Dual GLP-1/GIP Agonist)
Brand names: Mounjaro® (diabetes indication), Zepbound® (weight management indication)
Manufacturer: Eli Lilly
Tirzepatide represents an evolution in receptor targeting—a 39-amino-acid peptide that activates both GLP-1 and GIP (glucose-dependent insulinotropic polypeptide) receptors. This dual mechanism appears to enhance metabolic effects beyond single-receptor activation.
The SURMOUNT clinical trial program evaluated tirzepatide across multiple Phase 3 studies published in The New England Journal of Medicine (PMID: 35658024). FDA granted approval for type 2 diabetes (2022) and chronic weight management (2023).
Retatrutide (Triple GLP-1/GIP/Glucagon Agonist)
Status: Phase 3 clinical trials (anticipated FDA submission 2025-2026)
Manufacturer: Eli Lilly
Retatrutide adds glucagon receptor activation to the GLP-1/GIP mechanism—a triple agonist approach. Phase 2 data has been published in The New England Journal of Medicine (PMID: 37351564), generating significant scientific interest in the triple-agonist approach.
The glucagon component may enhance hepatic lipid oxidation and energy expenditure, though the full mechanism remains under investigation.
Global Regulatory Landscape
The US regulatory framework creates a specific market structure for these compounds:
- FDA approval required for therapeutic claims and prescribing
- Patent protection blocks generic manufacturing for approved indications
- Research use classification exists for compounds used in non-therapeutic laboratory settings
Regulatory classifications vary across jurisdictions. Researchers should consult applicable regulations in their region when procuring compounds for laboratory use.
Related Compounds in Development
The metabolic peptide field extends beyond the current FDA-approved options:
Cagrilintide — An amylin analog. Novo Nordisk is developing CagriSema, a combination with semaglutide, currently in Phase 3 clinical trials.
Survodutide — A dual GLP-1/glucagon agonist from Boehringer Ingelheim with a different receptor activation profile than tirzepatide.
Mazdutide — Developed by Innovent Biologics. Phase 3 results have been published; this compound may enter additional markets pending regulatory review.
Research Applications
GLP receptor agonists serve multiple in-vitro research purposes:
- Receptor binding studies — Characterizing GLP-1, GIP, and glucagon receptor pharmacology
- Cell signaling research — Investigating downstream pathways including cAMP, insulin secretion, and appetite-related gene expression
- Comparative analysis — Evaluating single vs. dual vs. triple agonist mechanisms
- Stability studies — Testing formulation and storage conditions
- Analytical method development — Establishing HPLC, MS, and other detection protocols
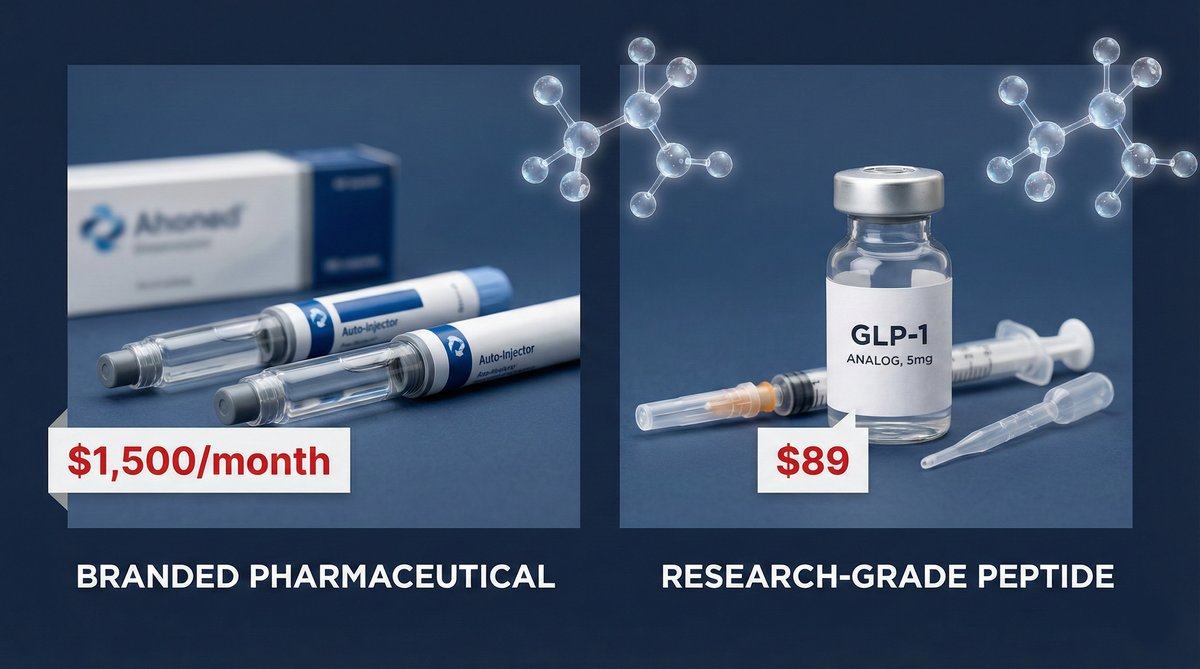
Epiq Aminos provides research-grade GLP agonists with purity exceeding 99%, verified by third-party HPLC and mass spectrometry analysis. Every batch ships with a Certificate of Analysis documenting molecular identity and purity.
Available Research Compounds
- GLP-1 S — Semaglutide analog for in-vitro research, purity exceeding 99%, third-party verified
- GLP-2 TZ — Tirzepatide analog for in-vitro research, purity exceeding 99%
- GLP-3 RT — Retatrutide analog available for research applications
- Cagrilintide — Amylin analog for in-vitro research
- Survodutide — Dual agonist for in-vitro research
Key Published Literature
Researchers interested in the clinical pharmacology of GLP receptor agonists may consult the following published trials:
- STEP Program (semaglutide) — NEJM, PMID: 33567185
- SURMOUNT Program (tirzepatide) — NEJM, PMID: 35658024
- Retatrutide Phase 2 — NEJM, PMID: 37351564
All products are intended for in-vitro research and educational purposes only. Not for human or veterinary use. These compounds are not drugs and are not intended for the treatment, prevention, or cure of any disease or condition. Consult published literature for clinical pharmacology information.